European CML Registry
European Registry of chronic myelogenous leukemia patients in failure
after imatinib therapy
Author: F. Guilhot, Université de Poitiers, France, Information Letter No.5, August 2008
Although imatinib is now the first line therapy for chronic myelogenous leukemia patients, experimental and clinical studies suggest that imatinib as a single drug might not be sufficient to eradicate Ph-positive stem cells.
Moreover, a concern with any single agent administered chronically is that resistant clones may emerge. Molecular studies of resistant leukemia cells isolated from patients have implicated BCR-ABL kinase domain point mutation as the most common mechanism of resistance. However, other mechanisms of resistance such as drug efflux, drug metabolism, BCR-ABL amplification have also been described. Current knowledge of the efficacy of imatinib is based on the results of the large IRIS trial. In this trial the risk of progression
after 5 years of therapy is near zero for patients who achieved a complete and sustained cytogenetic response. However, the true rate of failure patients treated with imatinib as a single agent is currently unknown. Knowledge on this subject is essential since no information is available concerning the true rate of failure outside clinical trials or unpublished studies.
Based on the considerations listed above, reasons for stopping imatinib could be multiple and the therapeutic decisions following imatinib failure are not well described. Thus, the purpose of the imatinib failure patient (IFP) registry is to
- collect data of imatinib failure CML patients in Europe
- document all subcategories of failure
- collect information on the treatment which was proposed
- collect information on the risk profile.
Therefor, the research questions are as followed:
- How many patients experience: Lack of efficacy (failure) under imatinib therapy? Toxicity leading to discontinuation of imatinib?
- What are the clinical and biological profiles of failure patients who experienced failure or toxicity?
- What was the dose and duration of treatment by imatinib until failure or toxicity was recorded?
- What was the medical decision after discontinuation of imatinib therapy?
Mechanisms of resistance are not detailed, and the different categories of mutations are not recorded in this registry. The IFP registry is organized as a sub-registry under the EL N-CML Registry (WP 4). IFP registry is supported by a grant from the 6th European Framework
program and by Novartis Pharma. French authorities approved this study in accordance with the European Community and Helsinki protocol.
As of December 2007, 567 patients (M 55%; F 45%) were recorded (Fig. 1).
Fig. 1: Number of patients
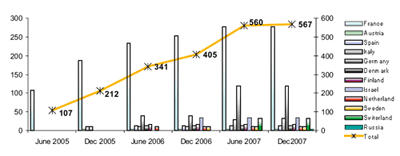
Median age at diagnosis was 51 (range 12-80 years). Fifty seven percent were never included in prospective studies at the time of failure. Reason for registration was no or unsatisfactory response in 28% of cases, loss of responses and progression to accelerated phase or blast crisis (23%) or discontinuation because of toxicity (17%) (Tab. 1).
| Reason for registration | [N] | % |
|---|---|---|
| Total | 567 | 100 |
| No CHR (after 3 month) | 12 | 2 |
| Not any CyR (after 6 month) | 56 | 10 |
| No MCyR (after 12 month) | 90 | 16 |
| Hematological relapse | 42 | 7 |
| Cytogenetic relapse or progression | 65 | 11 |
| Progression to accelerated phase or blast crisis | 130 | 23 |
| Toxicity (leading to discontinuation) | 95 | 17 |
| Miscellaneous | 77 | 14 |
End of 2007 the registry was activated in several participating centers of 12 European countries. The number of centers in Europe is constantly increasing and we are currently registering both new patients and the follow-up of patients with a target number of 1000 patients by the end of 2008.